Liquified vs compressed gases

Credit: stock.adobe.com
The British Aerosol Manufacturers Association (BAMA) explores a technical comparison between two aerosol spray processes
One question that is often asked of aerosol technologists is: “Why don’t you just use compressed air in all your products?” It is a perfectly reasonable question, given that any of the liquified propellants we use are flammable and expensive. Air is non-flammable (thank goodness) and is inexpensive.
Let’s compare some of the properties of compressed versus liquified propellants.
Liquified gases
- State: Liquified gases are gases that have been cooled to low temperatures, below their boiling point, to convert them into a liquid state.
- Storage: They are stored at the pressures needed to keep the gas in liquid form.
- Volume: Liquified gases occupy less volume compared to their gaseous state, making them easier to transport over long distances.
- Examples: Common examples include Propane, n-Butane, Iso-Butane and Dimethyl Ether (DME).
Compressed gases
- State: Compressed gases are gases that have been pressurised to reduce their volume but remain in a gaseous state.
- Storage: They are stored in high-pressure cylinders or tanks as gas.
- Volume: While compressed gases occupy less volume than their uncompressed state, they still take up more space compared to liquified gases.
- Examples: Common examples include Nitrogen and compressed air.
Let’s start with a bit of science. Boyle’s Law is a fundamental principle in physics and chemistry that describes the relationship between the pressure and volume of a gas at constant temperature. It states that the pressure of a given amount of gas is inversely proportional to its volume, provided the temperature remains constant. Mathematically, it can be expressed as: P×V= K where: P is the pressure of the gas, V is the volume of the gas and K is a constant for a given amount of gas at a constant temperature.
This means that if the volume of the gas decreases, its pressure increases and vice versa if the temperature does not change. For example, if you compress a gas into a smaller volume, the pressure will rise.
Let’s consider a compressed gas in an aerosol dispenser: every container has two phases, a liquid phase and a gaseous phase. When using a compressed gas, the headspace is pressurised at filling to a set pressure, typically around 100psig. As the product is used, the level of the liquid will fall and the headspace volume will increase.
If we apply Boyle’s Law and the volume the gas occupies doubles, the pressure will halve. It really is as simple as that.
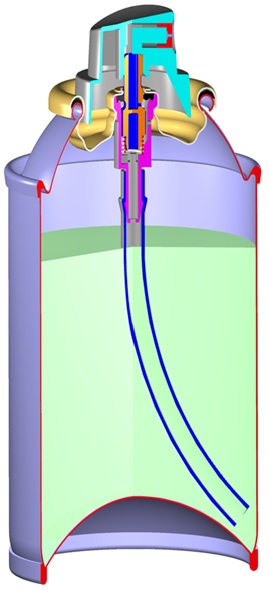
Compressed gas in an aerosol dispenser. Image provided by BAMA
Why is this a problem?
One of the key attributes of an aerosol dispenser is its ability to create a fine spray, but more importantly, a consistent spray through the pack life.
It does this partially by the valve and actuator technology, but also the propellant. If the pressure halves for every doubling of the headspace volume, then the spray will deteriorate.
Think of a garden sprinkler. If the tap is on full it will whizz around, spreading the water evenly over all your lawn. Reduce the pressure by closing the tap and the spread becomes less – the evenness of water distribution gets worse.
We solve this by using a liquified gas. As mentioned before, a liquified gas is one that has been cooled to below its boiling point and sealed into a cylinder. If it is then allowed to equilibrate to room temperature, some of the gas will turn to liquid – as in, it will boil. This will continue until the pressure in the headspace prevents any more gas changing to liquid at a characteristic pressure for a given gas or mixture of gases. This is referred to as the ‘Vapour Pressure.’
It is slightly more complicated. Imagine a glass of brandy in a large brandy glass. As you warm it in your hand some of the alcohol changes to gas. As it hits the cooler side of the glass, it turns back into a liquid. This cycle continues as long as you hold the glass (or drink the brandy). When you do this, you have created a dynamic equilibrium between the liquid and gaseous ethanol.
If we now think of an aerosol and carry out the same thought experiment, when you double the headspace volume, the pressure will still halve, but now we have disrupted the equilibrium between the gas and liquid forms of the propellant, so more propellant will change to gas to restore the vapour pressure. This is why liquified propellant aerosols are sometimes referred to as Constant Pressure Aerosols. By maintaining the pressure, we also maintain the spray characteristics through the pack life.
Other factors to consider are that the pressure in compressed gas propelled aerosols does not change significantly with temperature, so they are safer to use where high temperatures could be encountered and maintain their performance at low temperatures which makes them useful for products such as de-icers. Liquified propelled aerosols will see a significant rise in pressure with temperature and so every container carries a warning not to expose to temperatures exceeding 50°C.
The key questions the formulator must ask are: what spray characteristics apply to my product, and does it really matter if there is any change through pack life? One notable example is furniture polishes. If the product sprays reasonably well onto a surface it will work perfectly. A hairspray, for example, must have a specific particle size to work well, and must keep this through the pack life to maintain performance.
Some gases are readily soluble, such as Carbon Dioxide (CO2) and Nitrous Oxide (N2O). By dissolving the gas in the liquid of the product we create a limited reservoir of propellant, which gives some of the characteristics of a liquified gas.
Product applications
Which products are best suited to using compressed or soluble gases?
As mentioned before, products such as hairspray, flying insect killer and air fresheners that rely on a consistent particle size are much less suited to using compressed gases than, for example, furniture polishes or industrial products, such as brake and carburettor cleaner.
Hairspray relies on getting resin onto the hair strand and the liquid, once deposited, needs to migrate until it reaches where two hair strands touch. It then needs to dry to ‘fix’ the strands together. This is achieved by controlling the particle size, using the correct concentrate to propellant ratio and optimising the valve and actuator specification. As we have seen, one of the key issues with compressed gases is that the pressure drops over the life of the pack and hence the particle size will increase. This means as the spray hits the hair it will remain wet for an extended period, wetting the hair and causing the hair strands to soften and lose the hair style. One exception might be styling sprays intended for use with a hairdryer, where drying is accelerated.
Flying insect killers also rely on a similar principle. Insects breathe through their skin via small holes called spiracles so any solution containing insecticide must flow over the surface of the insect’s body until it reaches a spiracle where it can enter and kill the insect. The particle size must, therefore, be small enough to stay suspended in the air to allow the insect to fly through the cloud of droplets, but large enough so there is sufficient liquid on the surface to flow to the spiracle.
Air fresheners work by ensuring the droplets, containing fragrance, remain suspended in the air for an extended time. These droplets are then breathed in, and we perceive the fragrance. The ideal particle size is small enough to stay suspended, but not so small it is breathed into the deep lung. Particle sizes that are too large fall under gravity and hence reduce the efficacy.
However, there are several products where particle size, nor consistency are that important. Surface sprays such as furniture polishes, hard surface cleaners and disinfectant sprays just need to get the product onto the surface where it will be wiped with a cloth.
Industrial sprays are also good candidates for compressed gases. High pressures can be achieved so the ‘blast’ effect can be used with products such as brake and carburettor cleaners. Also, compressed gases are normally not flammable, so if the ingredients are also non-flammable it means that products can be formulated for use in hazardous areas.
BAMA provides a series of aerosol industry guides for manufacturing, R&D, recycling and waste, regulation standards, warehousing and more. Free for members, the guides can be downloaded by visiting www.bama.co.uk or emailing [email protected].






